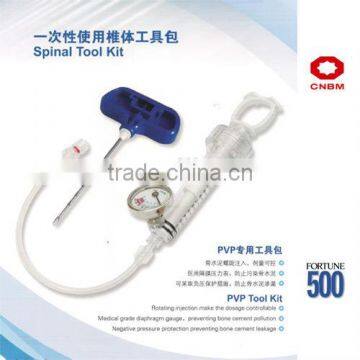
Percutaneous Vertebroplasty Kit
Specifications
1.MISS;2.Easy to operate,less operation time;Lower risk,less coplications;
3.Pain relief;
4.Reinforce the compressed verteb;

It is used to treat painful compression fractures caused by osteoporosis in the spine. It is to form a passage to restore the height of vertebrae, and to form a cavity for filling bone cement.
The operation of Percutaneous Vertebroplasty has the following features and benefits:
1. Minimally invasive approach to minimize the injury and bleeding;
2. Easy to operate, less operation time;
3. Regional anesthesia is applicable, lower risk, less complications;
4. Pain relief;
5. Reinforce the compressed vertebral body and help the patient to move;
6. Restore the height of fractured vertebrae.
Indications of Percutaneous Vertebroplasty:
1. Vertebral compression fractures caused by osteoporosis;
2. Vertebral compression caused by osteolytic pathology. Eg, vertebral hemangioma;
3. Sclerotin damage caused by malignant bone tumor with fracutre risk.
Contraindications of Percutaneous Vertebroplasty:
1. Patients with coagulation disorders;
2. Patients with back vertebral margin fracture and damage;
3. Patients with severe cardiopulmonary diseases;
4. Patients with cachexia of advanced tumor, who are intolerable to operation;
5. Patients predisposed to hemorrbage;
6. Patients with osteomyelitis, or existing systemic infection, or patients with inflammation at the injection site;
7. Bone protruded backwards, or tumor lumps in the rear side, which may involve the vertebral canal.

Send Inquiry to This Supplier
You May Also Like
-
Stainless Steel Implant & Interventional MaterialsUS$ 11 - 28MOQ: 100 Kilograms
-
CE ISO Approved PFNA Femur Trochanteric Nail, Interlocking Nail Orthopedic Implant China Factory, Intramedullary Nail Since 1958US$ 50 - 150MOQ: 1 Piece
-
Mini Fragment Instrument SetUS$ 4,000 - 5,000MOQ: 1 Set
-
Diesel Truck Body Part Lateral Plate 5301659-C4300US$ 1 - 25MOQ: 1 Set
-
Prosthetic Leg Orthotic Knee Joint Ring Lock, Drop LockNegotiableMOQ: 1 Set
-
Air Pump CAM Medical Orthopedic Walker Boot Medical Air Foot Orthopedic Fracture ShoesNegotiableMOQ: 100 Pairs
-
Medical Use of Disposable Infusion SetsUS$ 0.5 - 0.51MOQ: 1000 Bags
-
Titanium Hip Prosthesis Spine Pedicle ScrewsNegotiableMOQ: 1
-
Partial Knee SystemNegotiableMOQ: 1 Piece
-
4.0mm Orthopedic Cancellous Screw, Surgical Instruments, Orthopedic ImlpantUS$ 3 - 5MOQ: 1 Piece